If you follow news about weight loss medications, you have probably heard of Ozempic and Zepbound. Both drugs changed how doctors and patients think about treating obesity. But a newer drug called retatrutide is generating real excitement in the medical community, and for good reason. The clinical trial results are unlike anything we have seen before in pharmaceutical weight loss research.
Here is everything you need to know about retatrutide: what it is, how it works, what the trial data actually shows, and how it compares to the medications already on the market. And if you are currently exploring your options, our medically supervised weight loss program uses FDA-approved GLP-1 medications that are available right now, while retatrutide completes its approval process.
What Is Retatrutide?
Retatrutide, also known by its research name LY-3437943, is an investigational once-weekly injectable drug developed by Eli Lilly. It is classified as a triple hormone receptor agonist, which means it activates three separate hormone pathways in the body at the same time.
That is what makes it different from every weight loss medication that came before it. Most drugs in this class target one or two receptors. Retatrutide targets three.
In Phase 3 clinical trials completed in December 2025, retatrutide produced an average body weight reduction of 28.7% at the highest dose over 68 weeks. That is the largest weight loss result ever recorded in a late-stage pharmaceutical trial for an obesity drug.
How Retatrutide Works: The Triple-Receptor Mechanism
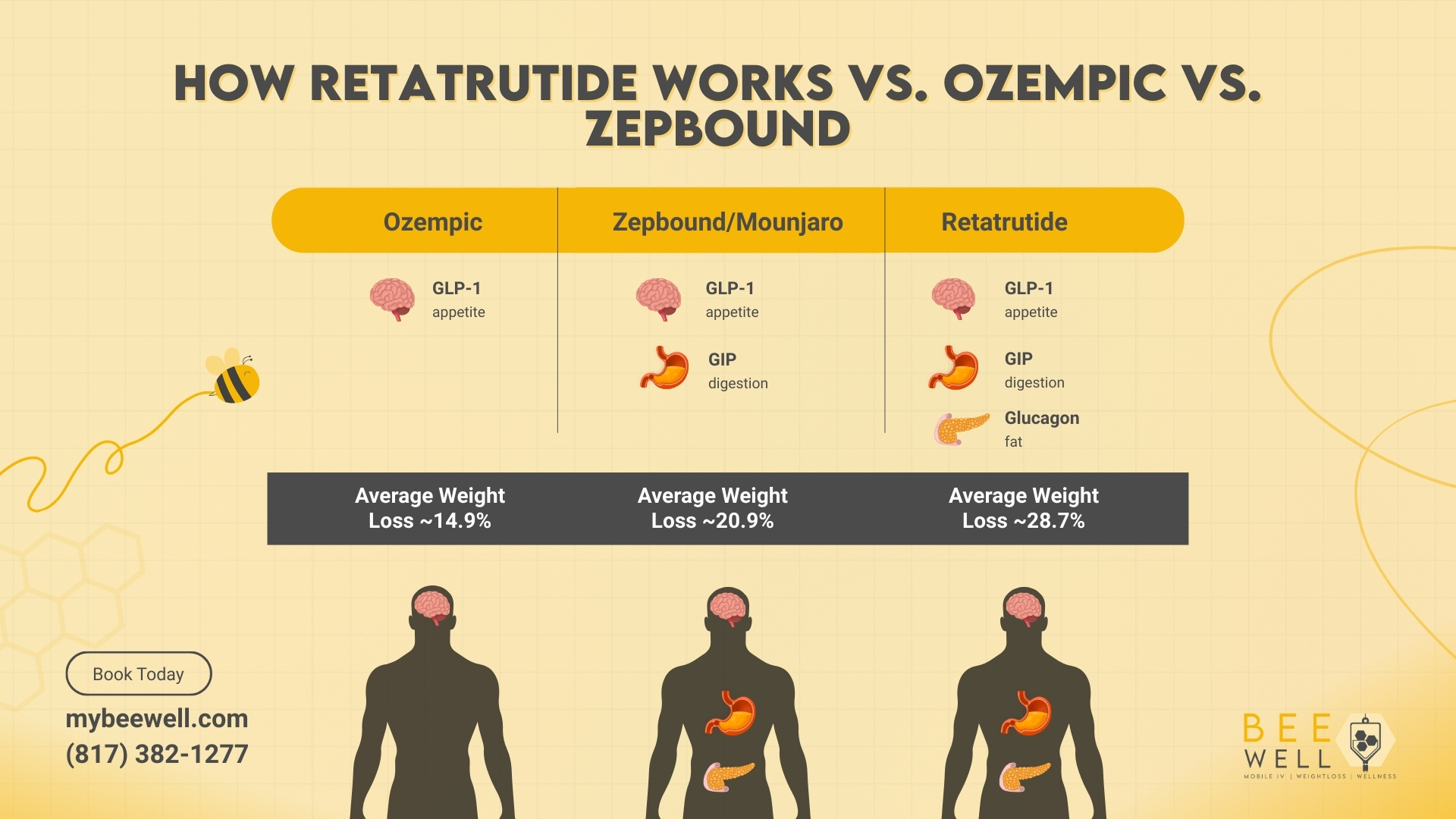
To understand why retatrutide produces such significant results, it helps to see how it compares to existing medications mechanically.
Semaglutide (Ozempic, Wegovy) targets one receptor: GLP-1. This receptor regulates appetite, slows gastric emptying, and stimulates insulin release.
Tirzepatide (Zepbound, Mounjaro) added a second receptor: GIP. Adding GIP enhances fat metabolism and improves insulin sensitivity, which is why tirzepatide outperforms semaglutide on weight loss.
Retatrutide adds a third receptor: glucagon.
What Each Receptor Does
GLP-1 Receptor Suppresses appetite, slows digestion, and stimulates insulin release. This is the same receptor targeted by Ozempic and Zepbound.
GIP Receptor Enhances insulin sensitivity, promotes fat burning, and amplifies GLP-1 activity. Also targeted by Zepbound.
Glucagon Receptor (unique to retatrutide). This is the key addition. Glucagon receptor activation increases energy expenditure, meaning the body burns more calories at rest. It also drives fat out of storage and into the bloodstream to be used as fuel, and produces significant liver fat reduction.
The result is a drug that reduces how much you eat, improves how your body processes sugar, and increases how many calories your body burns, all at once. That combination is why the trial results look so different from previous drugs.
Retatrutide Clinical Trial Results: The Data
Phase 3 TRIUMPH-4 Trial (December 2025)
The TRIUMPH-4 Phase 3 study enrolled 445 participants with obesity or overweight who also had knee osteoarthritis. Participants received once-weekly injections of retatrutide at either the 9mg or 12mg dose, or a placebo, over 68 weeks.
Average weight loss by dose:
- Retatrutide 12mg: 28.7% body weight reduction (approx. 71.2 lbs on average)
- Retatrutide 9mg: 26.4% body weight reduction
- Tirzepatide/Zepbound (Phase 3 comparison, not head-to-head): ~20.9%
- Semaglutide/Wegovy (Phase 3 comparison, not head-to-head): ~14.9%
- Placebo: ~2.1%
Note: These figures come from separate Phase 3 trials and are not direct head-to-head comparisons.
Beyond the average, the distribution of results is striking. At the 12mg dose:
- 58.6% of participants lost 25% or more of their body weight
- 39.4% of participants lost 30% or more
Those thresholds are typically associated with bariatric surgery. Retatrutide participants achieved them with a weekly injection.
The full TRIUMPH program enrolled more than 5,800 participants across multiple Phase 3 trials. Results from TRIUMPH-1, 2, and 3 are expected throughout 2026.
Benefits Beyond Weight Loss
The weight loss numbers get the most attention, but retatrutide’s triple-receptor mechanism produces a range of metabolic and systemic benefits that are worth understanding.
Cardiovascular Health
TRIUMPH-4 data showed meaningful reductions in non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein. The 12mg dose reduced systolic blood pressure by an average of 14.0 mmHg.
Joint Pain
Because TRIUMPH-4 specifically enrolled patients with knee osteoarthritis, the joint data is particularly detailed. Retatrutide reduced WOMAC pain scores by up to 4.5 points. At 68 weeks, 14.1% of participants in the 9mg group reported being completely pain-free, compared to 4.2% on placebo.
Blood Sugar Control
Phase 2 data showed HbA1c reductions of up to 2.02% at the 12mg dose. Ongoing TRANSCEND Phase 3 trials are evaluating retatrutide specifically for type 2 diabetes.
Liver Fat (MASLD)
A Phase 2 trial showed retatrutide reduced liver fat by 82%. Researchers described it as the most potent pharmacological liver fat reduction ever reported. Ongoing Phase 3 trials are evaluating this indication further.
Sleep Apnea and Kidney Disease
Separate ongoing TRIUMPH Phase 3 trials are evaluating retatrutide in patients with moderate-to-severe obstructive sleep apnea and in adults with chronic kidney disease and obesity.
At BeeWell, we have always taken a whole-body view of wellness. The breadth of retatrutide’s effects reflects something we see every day: when metabolic health improves, almost everything else follows. Energy improves, inflammation drops, joints feel better, and sleep quality goes up. Weight loss is part of the picture, not the whole picture.
Retatrutide Side Effects and Safety
The safety profile of retatrutide is broadly consistent with other drugs in the GLP-1 class. Most side effects are gastrointestinal and tend to diminish as the dose is gradually increased. There is one new side effect, however, that was not seen in earlier Phase 2 trials.
Reported Side Effects (Phase 3 TRIUMPH-4 Data)
| Side Effect | Rate at 12mg | Notes |
| Nausea | Up to 43.2% | Most common; improves with slow dose escalation |
| Diarrhea | Up to 34.7% | Common during escalation, diet adjustments help |
| Constipation | Up to 25.0% | Hydration and fiber reduce severity |
| Vomiting | Up to 20.9% | Less common than nausea |
| Dysesthesia | Up to 20.9% | See below |
| Decreased appetite | Common at higher doses | A therapeutic effect that requires monitoring at high levels |
Dysesthesia: The New Safety Signal
Dysesthesia is an abnormal sense of touch where normal sensations feel unusual, uncomfortable, or mildly painful. In TRIUMPH-4, it affected 8.8% of the 9mg group and 20.9% of the 12mg group, compared to just 0.7% on placebo.
This side effect was not observed in Phase 2 trials, and Eli Lilly’s research team is actively investigating its cause and long-term implications. It is one of the factors that could affect the FDA’s review timeline.
Discontinuation rates due to adverse events ranged from 12.2% to 18.2% across dose groups.
Contraindications
Retatrutide is not appropriate for people with a personal or family history of medullary thyroid carcinoma (MTC) or multiple endocrine neoplasia type 2 (MEN-2), or those with a history of pancreatitis. Anyone with these conditions should speak with their healthcare provider about alternatives.
Retatrutide vs. Ozempic vs. Zepbound: Full Comparison
| Feature | Ozempic/Wegovy | Zepbound/Mounjaro | Retatrutide |
| Receptor Targets | GLP-1 | GLP-1 + GIP | GLP-1 + GIP + Glucagon |
| Average Weight Loss | ~14.9% | ~20.9% | ~28.7% |
| Dosing Frequency | Once weekly | Once weekly | Once weekly |
| FDA Status | Approved (2021/2023) | Approved (2022/2023) | Phase 3, NDA expected 2026 |
| Liver Fat Reduction | Moderate | Significant | ~82% (Phase 2 data) |
| Systolic BP Reduction | Moderate | Moderate | 14.0 mmHg |
| Estimated Monthly Cost | $1,000 to $1,300 | $1,000 to $1,300 | ~$1,000 to $1,500 (projected) |
| Notable Side Effects | Nausea, GI | Nausea, GI | Nausea, GI, dysesthesia |
Can You Get Retatrutide Right Now?
This is the most common question we receive, and the answer is straightforward: no, you cannot legally obtain retatrutide outside of a clinical trial.
As of March 2026, retatrutide is not FDA-approved. It is an investigational drug that exists only within clinical trials sponsored by Eli Lilly.
How to Access Retatrutide Legally
The only safe and legal way to access retatrutide today is through enrollment in an active clinical trial. You can search for open trials at clinicaltrials.gov using the search terms “retatrutide” or “LY3437943.”
General eligibility criteria for most TRIUMPH trials include:
- BMI of 30 or higher, or BMI of 27 or higher with at least one weight-related condition
- No use of other weight loss medications in the prior 90 days
- No personal or family history of medullary thyroid carcinoma or MEN-2
- No recent pancreatitis, heart attack, or stroke (within the prior 90 days)
- No prior bariatric surgery
A note on online sales: The FDA has issued warnings about illegal websites selling substances that claim to be retatrutide. Anything purchased outside a clinical trial is unverified and potentially dangerous. Do not risk your health with unregulated products.
Retatrutide FDA Approval Timeline
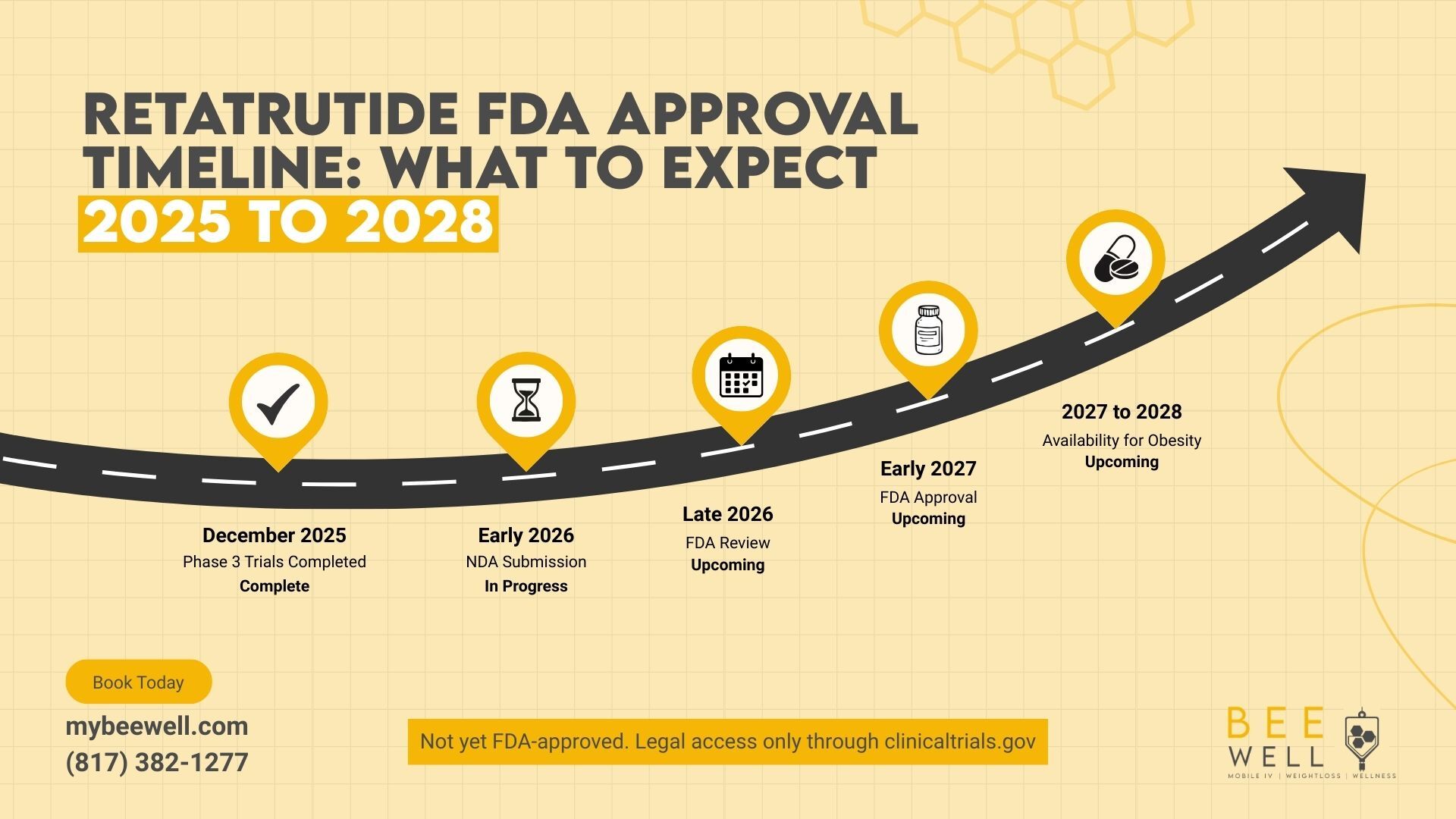
Here is a realistic timeline based on current Phase 3 data and standard FDA review processes.
December 2025 (Complete) TRIUMPH-4 Phase 3 topline results released. Average weight loss of 28.7% at 12mg confirmed.
January to May 2026 (In Progress): Remaining TRIUMPH Phase 3 trials (1, 2, and 3) are being completed. Seven additional Phase 3 readouts expected throughout 2026.
In late 2026, Eli Lilly is expected to submit a New Drug Application (NDA) to the FDA once full Phase 3 data is compiled and analyzed.
Early to Mid 2027 Standard FDA review period of 6 to 10 months. Priority Review designation is possible given the magnitude of the trial results, which could shorten this timeline to 6 months.
Mid to Late 2027: Potential FDA approval decision. The dysesthesia safety signal may prompt additional scrutiny or requests for supplemental neurological data.
2027 to 2028 Commercial launch. Initial availability is expected through specialty clinics, with broad pharmacy access following 3 to 12 months post-approval.
What You Can Do Right Now
Retatrutide is not yet available outside clinical trials, but that does not mean your weight loss journey needs to wait. At BeeWell, we offer medically supervised weight loss programs using FDA-approved GLP-1 medications that are available right now.
Visit us at mybeewell.com to learn more about our programs and start your consultation today.






